PharmaShots Weekly Snapshots (March 31, 2025 – April 04, 2025)
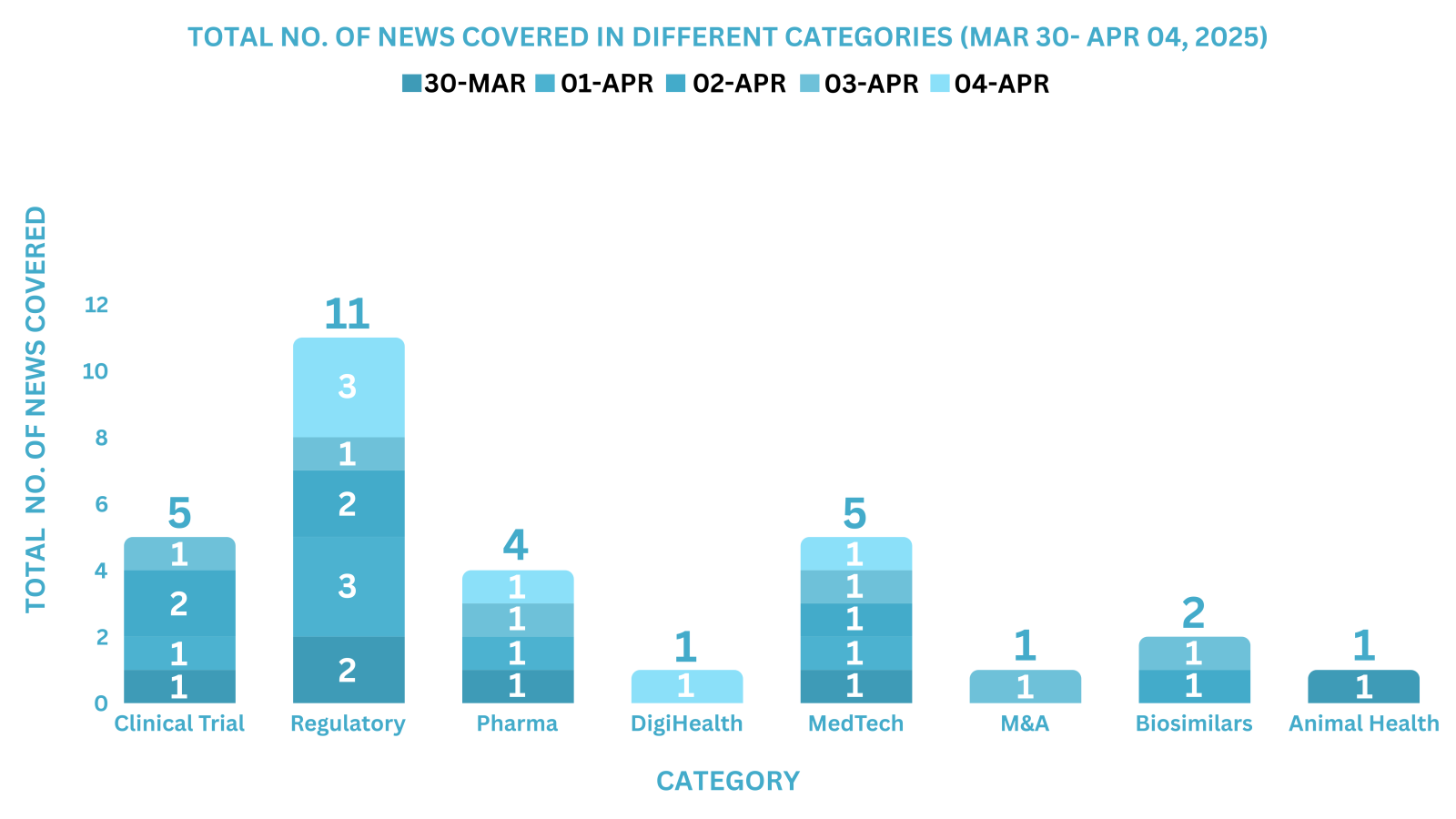
This week PharmaShots’ news was all about the updates on Clinical Trials, Regulatory, Pharma, MedTech, M&A, Biosimilars, Animal Health & DigiHealth. Check out our full report below:
 Eli Lilly Reports P-II (ALPACA) Trial Data of Lepodisiran for Heart Diseases
Eli Lilly Reports P-II (ALPACA) Trial Data of Lepodisiran for Heart Diseases
Read More: Eli Lilly
AstraZeneca Reports the Data from PURSUIT P-IIb Study Evaluating AZD0780
Read More: AstraZeneca
Axsome Therapeutics Reports Topline Data of P-III (PARADIGM) Trial of Solriamfetol for Major Depressive Disorder (MDD) regardless of Excessive Daytime Sleepiness (EDS)
Read More: Axsome Therapeutics
Daiichi Sankyo Reports the First Patient Dosing in P-III (DESTINY-Gastric05) Trial of Enhertu Regimen to Treat G/GEJ Cancer
Read More: Daiichi Sankyo
BridgeBio Oncology Therapeutics Reports the First Patient Dosing in P-I (KONQUER-101) Trial of BBO-11818 for Advanced Solid Tumors
Read More: BridgeBio Oncology Therapeutics
 BMS’ Opdivo Receives the CHMP’s Positive Opinion as a Neoadjuvant Treatment of Resectable NSCLC
BMS’ Opdivo Receives the CHMP’s Positive Opinion as a Neoadjuvant Treatment of Resectable NSCLC
Read More: BMS
AstraZeneca’s Calquence (Acalabrutinib) Plus Chemoimmunotherapy Receives the CHMP’s Positive Opinion for 1L Mantle Cell Lymphoma (MCL)
Read More: AstraZeneca
Genmab and Pfizer Secure the EC’s Approval for Tivdak (Tisotumab Vedotin) for Recurrent or Metastatic Cervical Cancer
Read More: Genmab and Pfizer
Alnylam Pharmaceuticals and Sanofi Report the US FDA’s Approval of Qfitlia (Fitusiran) to Treat Hemophilia A or B
Read More: Alnylam Pharmaceuticals and Sanofi
Pfizer Secures the EC’s Approval for Abrysvo (Respiratory Syncytial Virus Vaccine) for RSV Lower Respiratory Tract Disease
Read More: Pfizer
Apellis Pharmaceuticals Reports the US FDA’s sNDA Acceptance & Priority Review of Empaveli for C3G and Primary IC-MPGN
Read More: Apellis Pharmaceuticals
Averoa Receives the CHMP’s Positive Opinion for Xoanacyl to Treat Chronic Kidney Disease
Read More: Averoa
Novartis’ Vanrafia (Atrasentan) Receives the US FDA’s Accelerated Approval for Primary IgA Nephropathy (IgAN)
Read More: Novartis
Amgen’s Uplizna Receives the US FDA’s Approval for Immunoglobulin G4-related Disease (IgG4-RD)
Read More: Amgen
Daiichi Sankyo and AstraZeneca Report the EC’s Approval of Enhertu for HER2 Low/Ultralow Metastatic Breast Cancer (MBC)
Read More: Daiichi Sankyo and AstraZeneca
AstraZeneca Reports the EC’s Approval of Imfinzi Regimen as Perioperative Treatment of NSCLC
Read More: AstraZeneca
 Lexicon Enters a Global Licensing Agreement with Novo Nordisk for LX9851
Lexicon Enters a Global Licensing Agreement with Novo Nordisk for LX9851
Read More: Lexicon and Novo Nordisk
Ono Enters into an Option Agreement with Reborna Biosciences for RNA-Targeting Small Molecule to treat CNS Disorders
Read More: Ono and Reborna Biosciences
Sanofi Licenses an Undisclosed Nurix Therapeutics’ Drug Program for Autoimmune Diseases
Read More: Sanofi and Nurix Therapeutics
Eli Lilly Enters a ~$1.4B Licensing Agreement with Sangamo Therapeutics for its STAC-BBB Capsid
Read More: Eli Lilly and Sangamo Therapeutics
 Visby Medical Reports the US FDA’s De Novo Authorization of its Women's Sexual Health Test for Over-the-Counter Use
Visby Medical Reports the US FDA’s De Novo Authorization of its Women's Sexual Health Test for Over-the-Counter Use
Read More: Visby Medical
Echo IQ Enters into Strategic Partnership And Integration Agreements With ScImage And MedAxiom for EchoSolv AS
Read More: Echo IQ, ScImage and MedAxiom
The US FDA Grants 510(k) Clearance to Teleflex’s AC3 Range Intra-Aortic Balloon Pump (IABP) for Cardiac Patient Mobility
Read More: Teleflex
The US FDA Grants 510(k) Clearance to Medivis’ Spine Navigation Platform
Read More: Medivis
AVITA Medical Launches Cohealyx for Wound Healing in the US
Read More: AVITA Medical
 Siemens to Acquire Dotmatics for $5.1B, Strengthening its Position in Life Science Market
Siemens to Acquire Dotmatics for $5.1B, Strengthening its Position in Life Science Market
Read More: Siemens and Dotmatics
 Organon Acquires Regulatory and Commercial Rights of Tofidence IV Infusion (Biosimilar, Actemra) from Biogen in the US
Organon Acquires Regulatory and Commercial Rights of Tofidence IV Infusion (Biosimilar, Actemra) from Biogen in the US
Read More: Organon and Biogen
Accord Healthcare Reports the CHMP’s Positive Opinion on Osvyrti and Jubereq (Biosimilar, Prolia & Xgeva)
Read More: Accord Healthcare
 Spirited Paw Launches Calm Chewable Wafers for Dogs and Cats
Spirited Paw Launches Calm Chewable Wafers for Dogs and Cats
Read More: Spirited Paw
 NetraMark Join Forces with Worldwide Clinical Trials to Redefine Clinical Trial Design Using NetraAI Platform
NetraMark Join Forces with Worldwide Clinical Trials to Redefine Clinical Trial Design Using NetraAI Platform
Read More: NetraMark and Worldwide Clinical Trials
Related Post: PharmaShots Weekly Snapshots (March 24, 2025 – March 28, 2025)
Tags

Ridhi is an avid secondary researcher who follows trends in the biopharmaceutical and healthcare sectors to curate engaging content for the global audience. She works as a news editor at PharmaShots and loves to read books and explore new destinations.